Sudden Cardiac Death (SCD) – Risk Stratification and Prediction with Molecular Biomarkers
Sudden Cardiac Death (SCD) – Risk Stratification and Prediction with Molecular Biomarkers
Low Teck Yew and Junaida Osman
The heart is a biological pump that circulates blood throughout our bodies and thus supplying us with oxygen and nutrients. However, electrical stimuli that stimulate the heart can become disorganized, due to ventricular tachycardia or ventricular fibrillation [1], causing rapid death of brain cells and leading to sudden cardiac death (SCD) [2]. Stratification of clinical risks for SCD is therefore an important prevention measure. Traditionally, risk factors for cardiovascular diseases (CAD) and heart failures (HF) are accepted as predictors for SCD-related deaths [3]. These risk factors include (i) increased age, (ii) male gender, (iii) cigarette exposure, (iv) hypertension, (v) obesity, (vi) hypercholesterolemia, (vii) diabetes mellitus and (viii) family history and they have been incorporated into the US-based Framingham Risk Score and Europe-based HeartScore for estimating cardiovascular risks [4]. Nevertheless, these markers also preclude high-risk individuals without CAD symptoms [5].
Biomarkers are objective indicators of normal biological processes, pathogenic processes or pharmacological responses [6]. Ideally, a biomarker should be: (i) sensitive, (ii) specific, (iii) cost-effective, (iv) easily obtainable and (v) non-invasive [7]. Importantly, it should also be (vi) quantifiable, correlate well with the severity of disease conditions and (vii) able to offer early detection.
Since many SCD cases are heritable, early genetic studies apply the candidate gene approach to identify potentially meaningful genomic variants that are involved in various predisposing cardiac conditions. One genetic biomarker is SCN5A, which encodes the alpha subunit of the voltage-gated sodium channel Nav1.5 [8]. Any variations or mutations in SCN5A that affect the structure, function or expression of the sodium channel cause a delayed or persistent entry of sodium ions across the cell membrane, leading to arrhythmogenic syndromes and SCD. Potassium channel genes such as KCNH2 and KCNQ1 have also been found to carry mutations are present in long QT syndrome and were documented in SCD [9]. The cardiac ryanodine receptor (RyR2) is a calcium channel that regulates calcium ion release from the sarcoplasmic reticulum. A prominent RYR2 mutation that has been implicated in SCD is rs3766871 (p.Gly1886Ser), which is present in high prevalence in a molecular autopsy study involving 173 SCD cases, whereby rs3766871 has been demonstrated to result in an increased calcium ion oscillation in the cell and has been postulated to cause diastolic calcium ion leak [10]. As such, it is not surprising that the rs3766871 variant was found to be associated with an almost 2-fold increased risk of SCD. Mutations in other cardiac-related genes that have been implicated in SCD include: MYBPC3, which encodes cardiac myosin binding protein C [11]; ACE, which encodes angiotensin converting enzyme [12] and PKP2, which encodes plakophilin 2 [13].
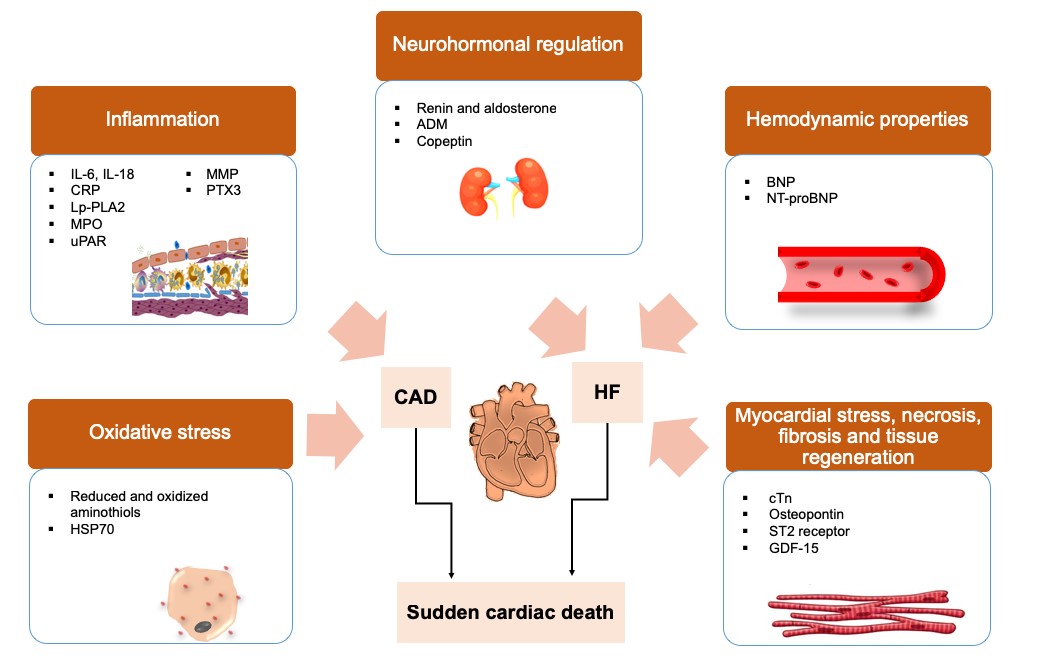
On the other hand, proteins are routinely used in clinical diagnostics. Biofluids are rich and non-invasive sources of circulating proteins that can provide quantifiable readout as biomarkers. Protein biomarkers for cardiac disorders often reflect the underlying pathophysiological processes in CAD or HF, two major causes of SCD. Oxidative stress represents an initiating event in CAD. It can be assessed by quantifying the levels of plasma aminothiol antioxidants such as cysteine and glutathione and their oxidized counterparts, i.e. cystine and glutathione disulfide [14]. Heat shock proteins (HSPs) are also upregulated during oxidative stress [15]. Among the interleukins, IL-6 and IL-18 are established as inflammatory biomarkers that are associated with CAD. On the other hand, C-reactive protein (CRP) is an acute-phase protein that is secreted by the liver in response to circulating levels of IL-6, IL-1 and TNF-α during the atherosclerotic process [16]. It is also a biomarker for CAD and SCD and can be measured with a high sensitivity CRP (hs-CRP) assay at sub-clinical levels (0.5 to 10 mg/L).
HF occurs when the heart is unable to pump sufficient blood to supply nutrients and oxygen. Elevated renin and aldosterone levels were found to be associated with HF and SCD in the LURIC study [17]. The adrenergic nervous system (ANS) system can also become dysregulated in HF. For example, adrenomedullin (ADM) is a peptide hormone with natriuretic, vasodilatory and hypotensive effects [18] and its concentrations were shown to become elevated in chronic HF [19]. During cardiac hemodynamic stress, natriuretic peptides (NP), i.e. BNP or NT-proBNP are secreted. Besides being capable of lowering blood pressure, NPs carry natriuretic, diuretic and kaliuretic properties [20]. NT-proBNP has been reported as an independent risk marker for SCD [21]. Tropomyosin interacts with cardiac troponin (cTnC, cTnI and cTnT), forming the troponin-tropomyosin complex that is responsible for cardiac muscle contraction. During myocardial stress, degeneration of the actin and myosin filaments results in the release of cTn into plasma. Therefore, cTnT and cTnI, being unique to the heart, are specific markers for myocardial damage. Both cTn and high sensitivity cTn (hs-cTn) assays have been used as predictors of mortality in both CAD and HF [22].
SCD is a fatal disease that has a very complex etiology. Although a number of risk factors and biomarkers have been used for diagnostics, prognostics and risk stratification for SCD, these biomarkers need to be further evaluated with larger and better-defined cohorts.
[1] Havmoller, R., Chugh, S.S., Plasma Biomarkers for Prediction of Sudden Cardiac Death: Another Piece of the Risk Stratification Puzzle? Circ. Arrhythmia Electrophysiol. 2012, 5, 237–243.
[2] Fukuda, K., Kanazawa, H., Aizawa, Y., Ardell, J.L., Shivkumar, K., Cardiac Innervation and Sudden Cardiac Death. Circ. Res. 2015, 116, 2005–2019.
[3] Adabag, A.S., Luepker, R. V, Roger, V.L., Gersh, B.J., Sudden cardiac death: epidemiology and risk factors. Nat. Publ. Gr. 2010, 7, 216–2253.
[4] D’Agostino, R.B., Vasan, R.S., Pencina, M.J., Wolf, P.A., et al., General Cardiovascular Risk Profile for Use in Primary Care. Circulation 2008, 117, 743–753.
[5] Myerburg, R.J., Goldberger, J.J., Sudden Cardiac Arrest Risk Assessment. JAMA Cardiol. 2017, 2, 689.
[6] Strimbu, K., Tavel, J.A., What are biomarkers? Curr. Opin. HIV AIDS 2010, 5, 463–6.
[7] Institute of Medicine (US) Forum on Drug Discovery D and T. Qualifying Biomarkers. National Academies Press (US); 2008 [cited 2018 Oct 22]; Available from: https://www.ncbi.nlm.nih.gov/books/NBK4041/ n.d.
[8] Marcsa, B., Dénes, R., Vörös, K., Rácz, G., et al., A Common Polymorphism of the Human Cardiac Sodium Channel Alpha Subunit (SCN5A) Gene Is Associated with Sudden Cardiac Death in Chronic Ischemic Heart Disease. PLoS One 2015, 10, e0132137.
[9] Winkel, B.G., Larsen, M.K., Berge, K.E., Leren, T.P., et al., The prevalence of mutations in KCNQ1, KCNH2, and SCN5A in an unselected national cohort of young sudden unexplained death cases. J. Cardiovasc. Electrophysiol. 2012, 23, 1092–8.
[10] Tester DJ, Medeiros-Domingo A, Will ML, Haglund CM, Ackerman MJ. Cardiac channel molecular autopsy: insights from 173 consecutive cases of autopsy-negative sudden unexplained death referred for postmortem genetic testing. Mayo Clin Proc. 2012;87:524–39 A n.d.
[11] Calore C, De Bortoli M, Romualdi C, Lorenzon A, Angelini A, Basso C, et al. A founder MYBPC3 mutation results in HCM with a high risk of sudden death after the fourth decade of life. J Med Genet. 2015;52:338–47 Available from: https://jmg.bmj.com/content n.d.
[12] Chen, Y.-H., Liu, J.-M., Hsu, R.-J., Hu, S.-C., et al., Angiotensin converting enzyme DD genotype is associated with acute coronary syndrome severity and sudden cardiac death in Taiwan: a case-control emergency room study. BMC Cardiovasc. Disord. 2012, 12, 6.
[13] Lahtinen, A.M., Havulinna, A.S., Noseworthy, P.A., Jula, A., et al., Prevalence of arrhythmia-associated gene mutations and risk of sudden cardiac death in the Finnish population. Ann. Med. 2013, 45, 328–35.
[14] Jones, D.P., Liang, Y., Measuring the poise of thiol/disulfide couples in vivo. Free Radic. Biol. Med. 2009, 47, 1329–1338.
[15] Kalmar, B., Greensmith, L., Induction of heat shock proteins for protection against oxidative stress. Adv. Drug Deliv. Rev. 2009, 61, 310–318.
[16] Yudkin, J.S., Stehouwer, C.D.A., Emeis, J.J., Coppack, S.W., C-Reactive Protein in Healthy Subjects: Associations With Obesity, Insulin Resistance, and Endothelial Dysfunction. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 972–978.
[17] Tomaschitz, A., Pilz, S., Ritz, E., Morganti, A., et al., Associations of plasma renin with 10-year cardiovascular mortality, sudden cardiac death, and death due to heart failure. Eur. Heart J. 2011, 32, 2642–2649.
[18] Peacock, W.F., Novel biomarkers in acute heart failure: MR-pro-adrenomedullin. Clin. Chem. Lab. Med. 2014, 52, 1433–5.
[19] Jougasaki, M., Rodeheffer, R.J., Redfield, M.M., Yamamoto, K., et al., Cardiac secretion of adrenomedullin in human heart failure. J. Clin. Invest. 1996, 97, 2370–6.
[20] Pandit, K., Mukhopadhyay, P., Ghosh, S., Chowdhury, S., Natriuretic peptides: Diagnostic and therapeutic use. Indian J. Endocrinol. Metab. 2011, 15 Suppl 4, S345-53.
[21] Korngold, E.C., Januzzi, J.L., Gantzer, M. Lou, Moorthy, M.V., et al., Amino-Terminal Pro-B-Type Natriuretic Peptide and High-Sensitivity C-Reactive Protein as Predictors of Sudden Cardiac Death Among Women. Circulation 2009, 119, 2868–2876.
[22] Everett, B.M., Brooks, M.M., Vlachos, H.E.A., Chaitman, B.R., et al., Troponin and Cardiac Events in Stable Ischemic Heart Disease and Diabetes. N. Engl. J. Med. 2015, 373, 610–620.

